Innovation, Efficacy & Transparency
Our Team

Tobias Ripsom
Toby directs GALYNA’s plant powered solutions into the skin care universe. As the co-founder of Veritas Fine Cannabis, Colorado’s original craft cannabis, Toby had a front row seat to the first adult-use legalization of cannabis and hemp. In that era, the power of superiorly cultivated trees, and their magical budding flowers was all encompassing. Seeing this, he began to cobble together concepts friendlier to the masses yet still rooted in his gardens. It was clear, the game changing plant matter he processed everyday could also be a packaged good. After suffering from a lifetime of troubled skin and now aging out of its resiliency, he partnered with topical industry veterans to dabble with benchtop prototypes. Toby’s fascination with real results from illicit ingredients grew irresistible. Never satisfied with the proof behind claims, nor touch and feel of dispensary offerings versus what he saw in department store skin care, he set out to prove cannabinoids benefited every body.
Toby brought almost a decade of operational, financial and marketing management prowess from real estate related endeavors to the cultivation arena of Denver circa 2014. His company helped pioneer America’s first legal marketplace and legitimize the alternative vice through professional trust and best in class craftsmanship. Nearly a decade later, he is still learning daily how plant power can improve lives and bodies, then turning that knowledge into the next skin solution.
A one-time Nordic ski racer for both the Sun Valley Ski Education Foundation and University of New Hampshire Wildcats, he can still be found testing GALYNA formulations, both before, during and after endless high-altitude kilometers. A husband and father of two sons, he and his family reside in Denver, Colorado.

Marnie Peterson, PharmD, PhD
Dr. Marnie Peterson is an entrepreneur, innovator, and expert in infectious diseases. She has 25+ years of experience in the development and discovery of new therapeutics related to infectious diseases and wound healing. In 2016, she founded and became the CEO of Jackson, Wyoming-based Extherid Biosciences to focus on new approaches to preclinical research including live animal alternatives (ex vivo tissue models) that aim to accelerate and predict clinical outcomes. This unique approach to technology development led to research collaborations with the FDA, academics, and global companies in the pharmaceutical, medical device and cosmetic industries.
In 2020, Extherid merged with UK-based Perfectus Biomed and became Perfectus Biomed, LLC, which operated as a US-based subsidiary of Perfectus Biomed Group. The new entity brought together over 60 employees focused on improving lives through innovative science. Marnie was the US Managing Director and CSO of Perfectus Biomed Group which was uniquely positioned to support global clients from preclinical development and throughout the regulatory stage. In 2022, Perfectus Biomed Group was acquired by NAMSA to expand the expertise and service offerings in customized microbiology to the MedTech industry globally. Marnie continues serves as a consultant to the Center for Infectious Disease Research and Policy at the University of Minnesota and hosts a podcast Superbugs and You. Before establishing Extherid, Marnie was tenured faculty at the University of Minnesota in the Department of Experimental and Clinical Pharmacology for 11 years. At the University, she established a research program and led multimillion-dollar research projects with the National Institutes of Health, Department of Defense, and various foundations and industrial partners. In 2014, the Minnesota Business Magazine selected Marnie as a Change Agent finalist for the Leaders in Health Care Awards. She earned a PhD in Infection from the University of Birmingham Medical School, United Kingdom and PharmD and BS degrees from the University of Minnesota.
She has published over 50 scientific manuscripts in peer-reviewed journals including Nature and holds a significant patent portfolio for her discoveries, which serve as the basis for a clinical stage women’s health company, Hennepin Life Sciences that was licensed to Dare Biosience. Importantly, these patents, experiences and life lessons informed her role as CSO of GALYNA.
In 2014, the Minnesota Business Magazine selected Marnie as a Change Agent finalist for the Leaders in Health Care Awards. She earned a PhD in Infection from the University of Birmingham Medical School, United Kingdom and PharmD and BS degrees from the University of Minnesota.

The Future of Cannabinoid Science
GALYNA™ is the synthesis of institutional life sciences and legalized craft cannabis. Our team’s history of success includes 45+ years of organic skincare formulation among the biggest names in the industry, a cannabis brand’s original co-founding pioneer, and scientific testing and precision by an expert in the development of topical products.
How do we know it works?
We have spent years researching and developing advanced topical formulations, and running a vast range of specialist studies via an independent laboratory to demonstrate efficacy, skin rejuvenation and restoration to ensure the integrity of our research and findings.
We test for proven effectiveness using human skin samples ethically sourced from post-plastic surgeries. It is here, under the microscope and other biochemical studies, that we explore skin cell changes to develop effective solutions and guide GALYNA™ formulations into the future.
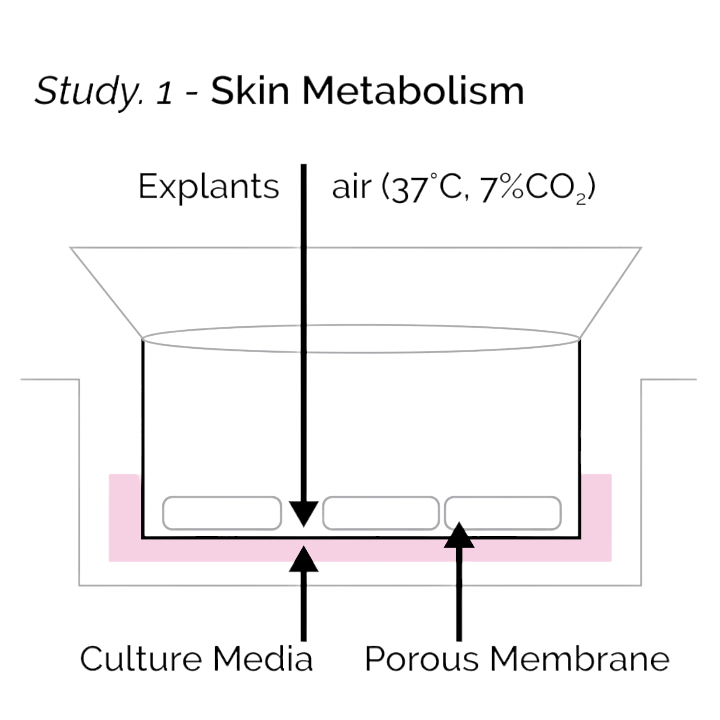
Study 1 – Skin Metabolism
Your skin is the largest organ of the body and a healthy metabolism is crucial to your human health. Skin metabolism is important in controlling regrowth and detoxification properties. Increased metabolic activity is widely defined as: managing rates of cell turnover and regeneration, collagen production, and repairing damage from UV exposure and aging.
What we Learned
Our proprietary formulations encourage positive biotransformation, and cellular regeneration within 24 hours. Through laboratory testing, we discovered that our formulations can enhance skin metabolism up to 50% more than untreated skin, in a non-irritating manner, and increase healing.
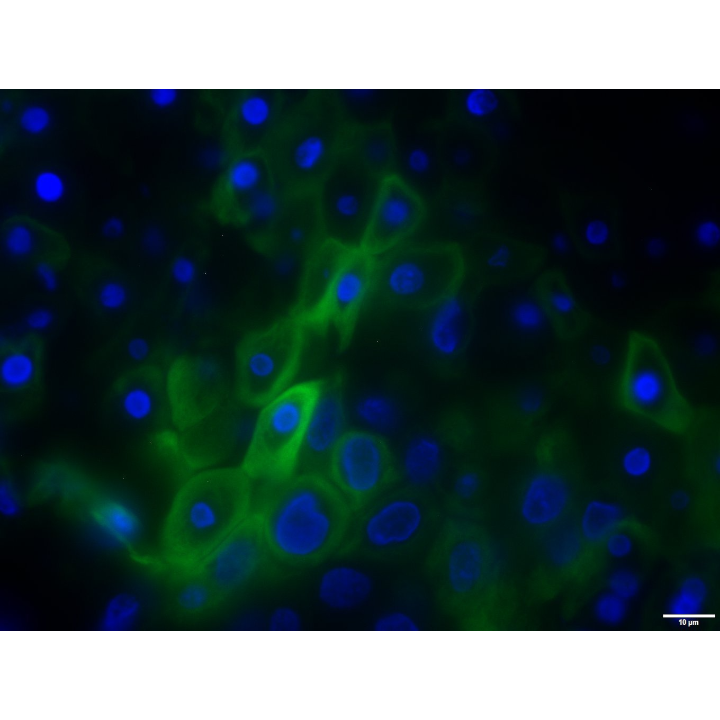
Study. 2 – Wound Healing
The study was led by our Chief Scientific Officer, whose previous experience specialized in prevention of the spread of infectious disease through open wounds and has conducted topical wound healing development studies for the Department of Defense, National Institute of Health and other multinational organizations.
For this study, we sourced human skin explants and created wound beds to treat with our cannabinoid-infused, lanolin-based Repair formulation. These samples were tracked and observed for a week at 63x magnification. Stimulation of wound healing and re-epithelialization (ie. the resurfacing of a wound with new epithelium) of the wound bed was noted by production of keratinocytes (K-17).
What We Learned
A one-time application of Repair stimulated the production of keratin 17 (K17) a cytokeratin important for epithelial migration and wound healing. The data shows that through skin penetration, our cannabinoid-infused formulations are able to reach the intended target receptors and promote wound healing via K17 expression.
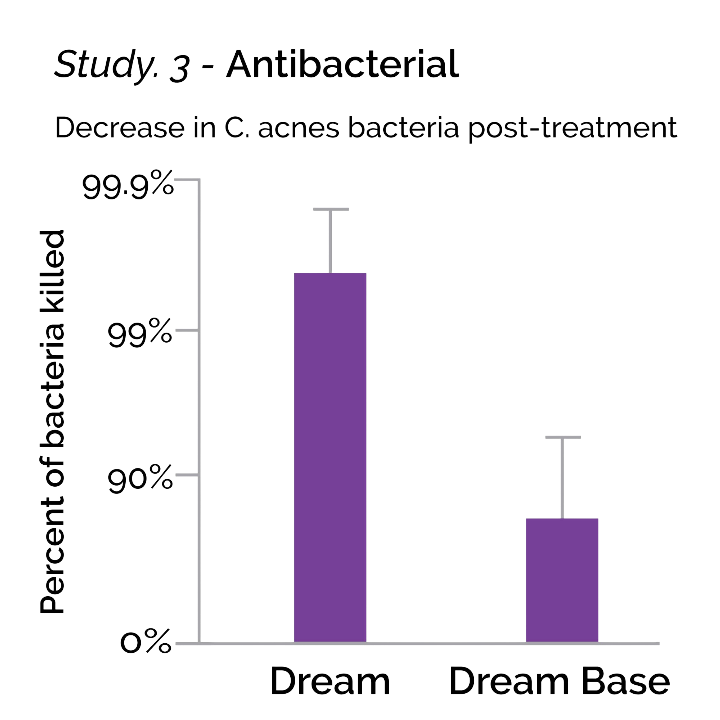
Study. 3 – Antibacterial
This study was conducted to determine the antibacterial/anti-acne effectiveness of GALYNA™ formulations against the bacteria, Cutibacterium acnes, which causes acne. Ex vivo human skin obtained via post-surgical elective surgery and under IRB protocol was prepared as 5mm explants. Cutibacterium acnes was inoculated to the surface for 2 hours incubation prior to treatment with Dream (cannabinoid-containing) and Dream base (no cannabinoids).
What We Learned
After 24h exposure and incubation, C. acnes bacterial densities were determined and a reduction in bacterial counts was logged. Cannabinoid-containing Dream formulation significantly reduced C. acnes compared to growth controls of C. acnes. Dream base composition (lacking cannabinoids) did not significantly reduce C. acnes.
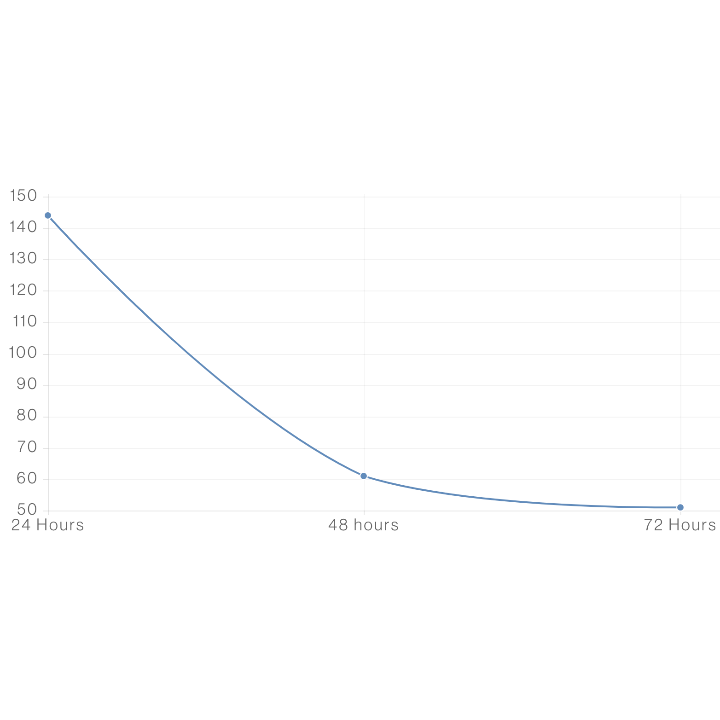
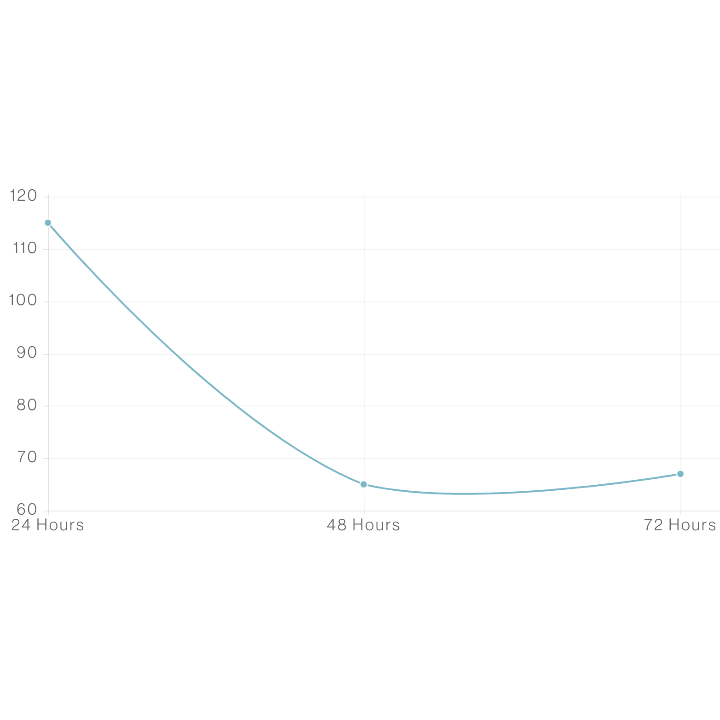
Study. 4 – Non-Irritation
For this study, our proprietary Dream and Repair formulations were all tested extensively using an adapted common standard irritation protocol (OECD 439), against commonly known irritants. A formulation can pass as non-irritating when 50% of cells are still viable after 24hrs of application. Viability of human skin after treatment with CBD containing formulations was assessed over three 24h continuous treatments of 10uL where formulations were washed off and re-applied at each time point.
The formulations were compared to controls that were non-irritating, moderate irritants, and strong irritants. This shows that all formulations tested did not decrease the viability of tissue in a manner that would indicate irritation. Our formulations passed with human skin cells remaining at above that mark, beyond twice that duration and only dipping below 50% near 72hrs.
What We Learned
The exposure time required to decrease viability to 50% is called the ET50. A longer ET50 means that skin tissue can be exposed to treatments for longer times before viability is decreased to half of the original. All formulations tested had an ET50 over 24hrs and were much higher than the mild irritant standard. Therefore, the formulations were classified as non-irritating substances.
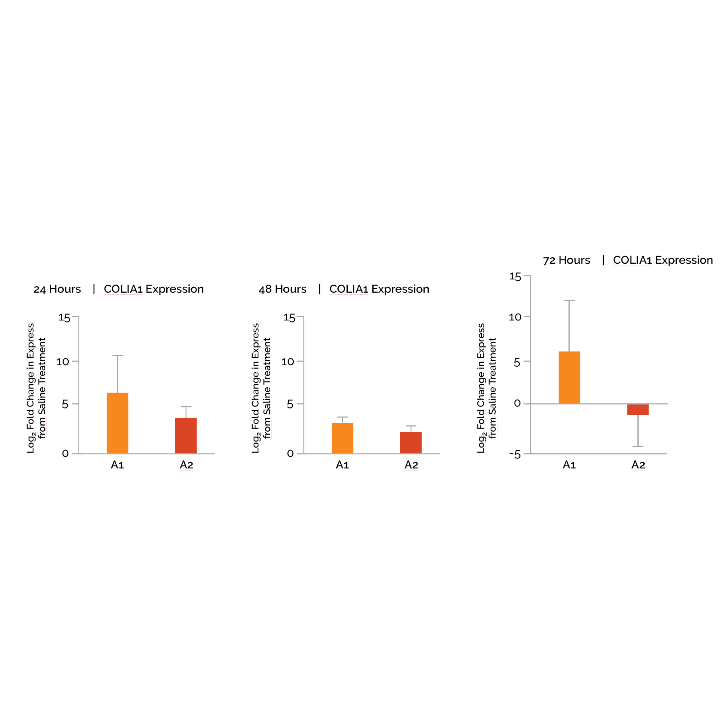
Study. 5 – Collagen Production
Our most recent study was conducted to assess the promotion of collagen production in Dream. As before, ex vivo human skin tissue was prepared and treated with Dream (A1) and the base (A2) formulation for analysis. Using reverse transcription-quantitative polymerase chain reaction (RT-qPCR), the most sensitive method for mRNA gene quantification, we were able to determine changes in collagen type I alpha 1 (COL1A1) gene expression in human skin following daily treatment with Dream. Type I collagen is the most abundant form of collagen in the human body.
What We Learned
After 72hrs, Dream (A1) showed a significant increase in COLIA1 gene expression, a Type I collagen, outperforming the base formula (A2) over 7- fold. Also in other studies not shown, the active formulation (A1) was non-irritating up to 72 hours as was demonstrated in the previous studies.

Our Mission:
The GALYNA™ mission is to provide science-based, all-natural, topical skincare products, by enhancing the full potential of cannabinoid science to help people take better care of their most essential barrier – the skin.

Our Vision:
Our advanced skincare formulations are the result of research backed science, and designed to be used in sync with your lifestyle and existing personal care regimen, for adults 21 years and older. We aim to be completely transparent about our research and results, and to continue expanding our knowledge for future product development.

Our People:
The GALYNA™ team boasts multiple doctorates, organic formulators from the biggest skin care laboratories and cannabis industry pioneers. We’ve enlisted the expertise of scientists, whose research work is focused on innovating, testing and developing bio-solutions that allow us to gain a greater understanding of the effects of various cannabinoids on the human body. With their help, we are continuously adapting and improving our formulas, to bring you the highest-quality products

Curious to Learn More?
Sign up to receive our latest scientific data, research reports and to participate in future product development.
Events
Coming Soon
Frequently Asked Questions
Products & Ingredients
General Knowledge
All humans, and many other animals, have an Endocannabinoid System (ECS), an endogenous system that regulates biological balance, or homeostasis.
The ECS consists of three key elements:
- Cannabinoid Receptors (CB1 and CB2),
- Endocannabinoids (chemical compounds produced by the body) and
- Metabolic Enzymes (produced to break down endocannabinoids).
Phytocannabinoids, such as CBD and THC, interact with the ECS by mimicking biological receptor responses.
CBD and THC are only two of 113+ identified chemical compounds, or cannabinoids, found in the cannabis plant. Both compounds interact with the naturally occurring human Endocannabinoid System.
CBD (Cannabidiol) is a major, non-psychotropic phytocannabinoid (i.e. plant produced compound that has no ‘mind-altering’ effects). CBD is produced in large concentrations in the cannabis plant species, and can also be found in other plants, such as hops.
THC (Tetrahydrocannabinol) is the compound in cannabis that is psychoactive, and when consumed or smoked, it is absorbed into the bloodstream and travels to the brain, attaching itself to the naturally-occurring endocannabinoid receptors located in the cerebral cortex, cerebellum, and basal ganglia. These are the parts of the brain responsible for thinking, memory, pleasure, coordination and movement.
To shop GALYNA’s Extra Strength (THC) products, please visit www.galynathc.com.
Our Company
Pronounced ‘Gah-lee-nah’, the word has several interesting meanings:
1) It is the name of the Balkan Goddess of Calm and Tranquility.
2) Galena is also the name of a highly sought after ore used in silver mining in the intermountain west like Aspen & Sun Valley. 3) It is the name of an eye make-up used to fend off the sun by ancient Egyptians.